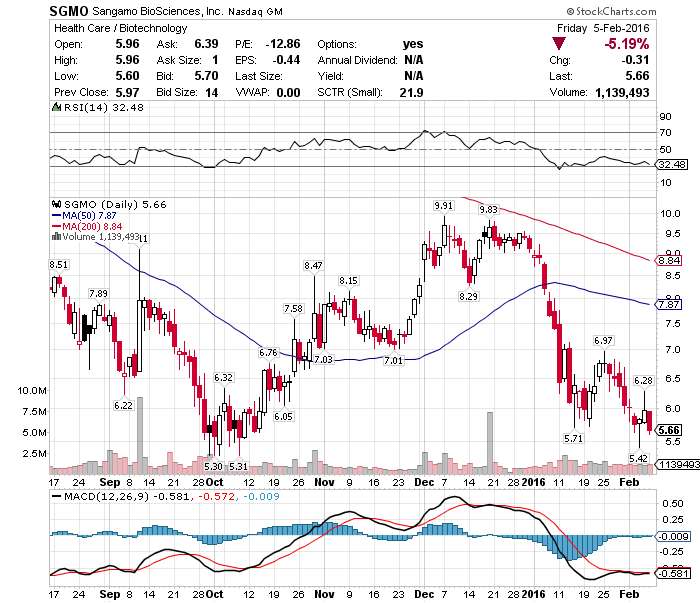
$SGMO today's scanner on FDA Clearance #PreMarketGainers @ $6.40 UP 13% TP @ $7
RICHMOND, Calif., Feb. 8, 2016 /PRNewswire/ -- Sangamo BioSciences, Inc. (SGMO), the leader in therapeutic genome editing, announced that the U.S. Food and Drug Administration (FDA) has cleared the Company's Investigational New Drug (IND) application for SB-318, a single treatment strategy intended to provide a life-long therapy for Mucopolysaccharidosis Type I (MPS I). The SB-318 IND application is now active and enables Sangamo to initiate a Phase 1/2 clinical study (SB-318-1502) designed to assess the safety, tolerability and potential efficacy of SB-318 in adults with varying severities of MPS I.
 SEE ALSO: Monday's Scanner
SEE ALSO: Monday's Scanner
RICHMOND, Calif., Feb. 8, 2016 /PRNewswire/ -- Sangamo BioSciences, Inc. (SGMO), the leader in therapeutic genome editing, announced that the U.S. Food and Drug Administration (FDA) has cleared the Company's Investigational New Drug (IND) application for SB-318, a single treatment strategy intended to provide a life-long therapy for Mucopolysaccharidosis Type I (MPS I).



No comments:
Post a Comment